Bio-based solutions for micronutrient extraction from wastewater
Preeti Chauhan*, Tanvi Salaria, Kanishika, Palki Sahib Kaur
Department of Biotechnology, Chandigarh College of Technology, CGC, Landran Mohali, Punjab, India-140307.
Abstract
Large volumes of wastewater produced as a result of extensive water consumption for urban, industrial, and agricultural uses cause many complications. An excessive amount of nutrients, particularly nitrogen (N) and phosphorus (P), in wastewater could lead to eutrophication in lakes and disturb the equilibrium of the ecosystem. Withdrawal of harmful metals and nutrients from wastewater to acceptable levels before it is discharged and used again is a crucial step in wastewater treatment. Nevertheless, the majority of traditional wastewater treatment systems rely on physical and chemical processes that are not costeffective for treating wastewater, particularly that from agriculture. Using wastewater as a source of nutrient elements like nitrogen and phosphate is an excellent option to develop microalgae for micronutrient extraction. The harvesting of microalgae to use as feedstock for biofuels like biogas will enhance its value. Despite the enthusiastic research that has been published on algae growth in wastewater and simultaneous micronutrient extraction, the challenge of the scale-up process still needs to be addressed. In this study, we present the use of microalgae to extract micronutrients. We notably discuss the techniques that have been shown to enhance the growth of microalgae and their interactions with other microbes as a bio-based solution to wastewater treatment. This study also emphasizes the potential of cocultivating microorganisms with microalgae, such as bacteria and yeast, for micronutrient extraction fromwastewater.
Keywords: Microalgae, wastewater treatment, micronutrient recovery, co-cultivation system, ecosystem balance
* Corresponding author: Dr. Preeti Chauhan
E-mail address: chauhanpreeti248@gmail.com
Article type: Mini-article
Received: March 22, 2025
Revised: April 14, 2025
Accepted: May 2, 2025
Please cite this article as: Chauhan P, Bio-based solutions for micronutrient extraction from wastewater, Biotechnol. kiosk, Vol 7, Issue 7, PP: 1-12 (2025); DOI: https://doi.org/10.37756/bk.25.7.7.1
Introduction
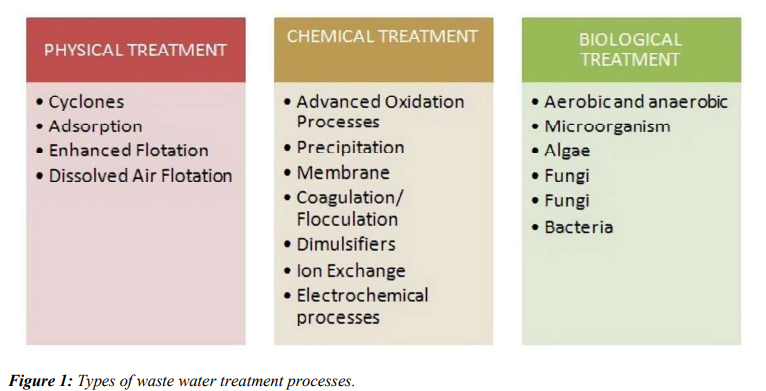
The recovery of micronutrients from waste water and bio-based waste is an area of interest due to the depletion of non-renewable resources and geopolitical issues. However, there is a need for a natural approach and environmentally friendly interventions due to growing environmental concerns [1]. In particular, the release of the nutrient-dense solution into the environment is endangering freshwater resources globally. The majority of treatment techniques involve chemical, physical, and biological processes as shown in (Figure 1). Microalgae have demonstrated efficacy in the management of solid and liquid waste associated with cattle, poultry, and aquaculture. Typically, these waste streams are high in micronutrients, which the microalgae absorb during growth [2]. Various biological and physicochemical technologies are available to recover the micronutrients [3]. Additionally, micronutrient recovery provides opportunities that extend beyond environmental preservation, such as the production of valuable products [4]. By treating wastewater, microalgae farming in residential or municipal wastewater (MWW) may be a practical way to improve environmental and economic sustainability [5]. Current studies have shown the use of microalgae to remove pesticides and medicinal compounds from wastewater created by industries and agriculture, in addition to their ability to withdraw nutrients from wastewater generated by WWTPs [6]. Microalgae are therefore an adaptable organism for treating numerous forms of liquid effluents emanating from various industrial and agricultural processes due to their capacity to thrive, assimilate, and resist hazardous conditions in wastewater [7]. This study examines wastewater treatment using native microalgae in combination with other systems, such as microalgae-built wetland systems, microalgaebacteria systems, and microalgae-yeast systems etc. together with an outlook on the future as well as challenges of microalgae in wastewater treatment.
Cellular composition of algae
The way that contaminants interact and are eliminated from wastewater is significantly influenced by the cellular makeup of algae. According to reports, the percentage of fat, carbohydrates, and protein in algal cells ranges from 5-23%, 7-9%, and 6-42%, respectively [8]. Microfibrillar exo-polysaccharides, which include functional groups like -COOH-, -OH-, -PO43-, -RSH, and SO42-, are part of the algal cell wall. These functional groups provide the cell wall with an anionic character. The content of nitrogen (N) and phosphorus (P) in the biomass may range from 5.4 to 8.7% and 0.7 to 1.1%, respectively, depending on the microalgal cellular composition. Thus, microalgae by themselves may be able to absorb or eliminate 43.6 and 5.6 mg/L of phosphorus (P) and nitrogen (N), respectively, from wastewater [9]. Furthermore, microalgae may be able to absorb chemicals from wastewater and help other microbes eliminate additional contaminants. Wastewater contains all the other micro-elements that are required [10].
Wastewater treatment by co-culturing
Some of the current studies have discussed the advantages of using microalgae consortiums over single-species cultures because microalgae have
been successfully used in the removal of nutrients from a variety of wastewaters, but maintaining microalgae monoculture in such processes is quite
difficult. Cultivating algae in a consortium has been thought to result in both cooperative and competitive relationships and can be best utilized to recover nutrients from waste water. The details of different consortia of algae are given in table 1. Co-cultivating algae with fungi is an important approach to effectively harvest microalgae because fungi have the potential to immobilize microalgae through mycelial interaction.
Researchers have shown that the biomass of the fungus-microalgae mixed culture was significantly higher than that of the pure culture [11]. Extracellular enzymes released by fungi have the ability to convert solid organic components into soluble nutrients and carbon dioxide, which facilitates their absorption by microalgae cells [12]. Co-cultivation of filamentous fungi with microalgae is a technique for effectively collecting microalgae by the process of co-pelletizing into fungal pellets.
Fungi-based flocculation technology is relatively potent compared to that of bacteria [9]. There are few studies in this field currently, and they are still in their early stages of development. The fungi microalgae combination shows enormous potential in treating wastewater sources such as municipal, industrial, pharmaceutical, agro-industrial wastewater, water derived from mining activity, aquaculture, etc. [13, 9]. The removal of contaminants from wastewater, including the biosorption of heavy metals, is assisted by the special surface characteristics of fungi and the abundance of degrading enzyme resources [14]. Algal-bacterial systems, fungal-algal systems, and multi-bacteria-algae systems are the three types of systems used in co-culture [15]. Quorum sensing (QS) can alter the motility of bacteria and important characteristics of the cell membrane surface, which can then impact sludge granulation, processes [16]. Tan et al. found that quorum sensing could enhance the production of adenosine triphosphate (ATP), boost the extracellular polymer secretion of sludge, and make it easier for aerobic granular sludge to develop [16].

Phosphorus Recovery
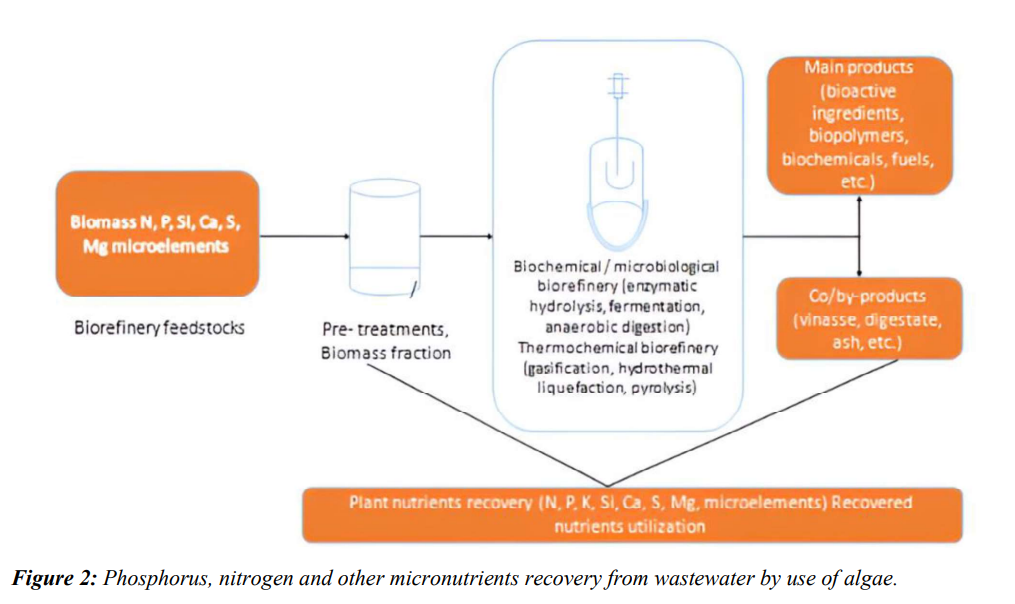
The goal of enhanced biological phosphorus removal/uptake from wastewater is to generate low-phosphorus effluent. Wastewaters contaminated with nitrogen (N), phosphorus (P), and other trace elements can be used by microalgae for their cell growth (Figure 2). Numerous studies have assessed the utilization of recovered phosphate products in recent decades, especially in relation to their application as fertilizer. The use of biobased phosphate added to the soil to increase soil fertility can be measured using a variety of techniques [17].
The greatest possible influence on phosphorus recovery can only be attained by selecting the optimal course of action given the specific circumstances. The environment, economy, and treatment typically have an impact on the recovery of phosphorus from sewage water, which means that many methods should be employed instead of a single one [18].
In open pond systems, microalgae have been utilized in wastewater treatment to eliminate organic contaminants and nutrients [19]. These days, a method of revolving algal biofilm (RAB) systems has been created to maximize light and nutrition conditions to boost microalgal development, hence increasing the effectiveness of nutrient removal from wastewater [20]. Wastewater-derived microalgae invade the RAB system and create an adherent biofilm on a vertical, partially submerged rotating belt. As the microalgae grow, cellular uptake of P and adsorption of other pollutants are made possible by intermittent submersion in wastewater, while gaseous exchange and penetration of light into the biofilm are made possible by exposure to the atmosphere. After that, the nutrient-rich microalgal biomass can be scraped off and used as feedstock for biofuel or other bioproducts, or as fertilizer [21].
Nitrogen Recovery
Numerous technologies, such as electrodialysis, ammonia stripping, struvite precipitation, and membrane-based technologies, have been introduced to enable the effectivet nitrogen recovery from waste water [22]. The majority of nitrogen in wastewater is found as total ammonia, as NH4+ or NH3. Ammonium oxidizing bacteria (AOB) convert ammonia to nitrite (nitritation) in aerobic nitrification, which is the conventional method of biological nitrogen removal (BNR). Denitrifiers use organic carbon as an electron donor to convert nitrate to dinitrogen gas [23]. Algae phosphorylate and absorb nitrogen to get nutrients from the growth medium. The process by which inorganic nitrogen (ammonium, nitrite, nitrate, ammonia, etc.) is transformed into its organic form i.e. building blocks of peptides, proteins, enzymes, chlorophylls, energy transfer molecules like adenosine diphosphate (ADP) and
adenosine triphosphate (ATP), and genetic materials like DNA and RNA is termed as nitrogen assimilation [24]. Nitrate and nitrite reductase, respectively, assist in the final reduction of nitrate and nitrite to ammonium during assimilation. With the aid of ATP, glutamate (Glu) and ammonium
are subsequently combined to form the intracellular amino acid glutamine. The method by which algae absorb nitrogen differs from that of bacteria. [25]. The nutrient uptake patterns of algae can be influenced by availability of nutrient for instance slow-growing micro algae, build up large nutrient stores by accumulating P and N during nutrient-rich times in order to grow during nutrient-depleted periods. These slow-growing algae maximize their potential to absorb carbon and minimize their nutrient needs by slowing growth rates when nutrients are scarce, taking advantage of the abundance of natural light [26]. The growth rate of both macroalgae and microalgae has been found to be limited by nitrogen. Additionally, it has been noticed that ammonium is frequently preferred nitrogen (N) source and is more easily absorbed by algae since it requires less energy for cells to ingest. The internal nitrogen content of the algal biomass is increased by higher nitrogen (N) concentrations, which facilitate the absorption of inorganic nitrogen (N) into amino acids and proteins. Therefore, ammonium-rich wastewater is a viable supply for algae growth. The crude protein content of algae growing in N-rich effluent was four times higher than that of seawater controls [27].
Challenges, limitations, and opportunities Although employing algae to treat wastewater is a viable and exciting approach, there are several restrictions before this technology reaches a mature state. The poor biomass production, high energy requirement, and economic viability of algae culture are critical challenges. Increasing production costs are associated with different methods of growth, harvesting, cell disruption, and extraction for algae-based wastewater treatment.
Genetic engineering-based methods for algal growth
A more comprehensive set of molecular tools will be needed to develop microalgae as a successful platform for tailored bioproducts. The algal
genome can be manipulated to yield the ideal amounts of the target products with the use of a variety of validated, reliable technologies. Using the wide range of engineering tool available, other microorganisms, such as yeast and bacteria, have been successfully cultivated to manufacture a highly diverse set of industrial products [28]. Because of recent developments in the creation and refinement of novel genetic components and
transformation methods, microalgae are gaining ground to become comparable to these other industrial microorganisms [29]. The area of microalgal genetics has advanced significantly over the last decade. The nuclear, mitochondrial, and chloroplast genomes of several microalgae have already been sequenced, and others are now being sequenced. Furthermore, databases of expressed sequence tags (EST) have been established. The green alga Chlamydomonas reinhardtii has traditionally been the subject of the majority of molecular and genetic phycological studies. Consequently, most of the gene knockdown and transgenic expression methods have been developed specifically for this species [30].
Algal treatments, on the other hand, are a useful method for treating a range of industrial wastewaters. With the help of particular algal species, microalgae can be utilized to remediate organic contaminants and treat pollutants in water. Additionally, the process of treating microalgae results in biomass that may be used to make biofuel and has the added advantage of absorbing CO2 [31]. Through these kinds of treatment procedures, algal cultures can produce valuable compounds that lower atmospheric CO2 levels and biofuels, thus providing solutions to both economic and environmental issues. Produced Water (PW) has been utilized to preserve freshwater supplies in response to rising demands for water resources, particularly in desert areas where freshwater is scarce. Studies on the application of algae for PW treatment found that it is not properly developed yet.
The majority of the interactions between bacteria and microalgae are species-specific, according to the microbial consortium.
1. Improved power generation
2. Treatment of wastewater and nutrients recovery 3. Cultivate algal biomass for the production of biofuel
4. Productions of CO2, O2, and
5. Maintain an appropriate ecological symbiotic relationship.
Microalga have been genetically engineered to enhance their ability to use sunlight efficiently in laboratory trails.
Potential, limitations and future of algal wastewater biofuel production
Most conventional and edible crops are used in the production of first-generation biofuels, such as biodiesel, bioethanol, and biobutanol. A renewable resource for the production of biofuels including biohydrogen, bioethanol, biogas, and biodiesel as well as other value-added goods like vitamins and antibiotics is algal biomass [32]. Biomethane and biohydrogen are regarded as co-products, whereas bioethanol and biodiesel are classified as major
products. Algal biomass can also be used for environmental remediation and the production of bioelectricity. The use of numerous microalgal species, including Scenedesmus sp., Actinastrum sp., Heynigia sp., Chlorella sp., Botryococcus, etc., has been suggested [33]. Microalgae have a high potential to be a viable, sustainable growing medium for biofuel feedstock due to their capacity to grow well under specific wastewater conditions, as previously
mentioned. Numerous studies on microalgae growing in wastewater have shown high biomass productivities and, in certain circumstances, high lipid productivities [34]. These findings indicate that there is potential for producing biofuel at a reasonable cost by utilizing these high nutrient supplies. Nonetheless, it is necessary to address certain constraints.
Metabolic editing
As mentioned earlier microalgae have a high potential to be a viable, sustainable growing medium for biofuel feedstock due to their capacity to grow well under specific wastewater conditions. Two major obstacles to the commercial production of high-value microalgal pigments are high culture costs and limited pigment yield [35]. Microalgae are thought to be promising sustainable feedstocks since they can directly convert CO2 into chemicals and fuels. However, the viability of a microalgal bioprocess from an economic standpoint is debatable. Therefore, there are numerous attempts to use metabolic engineering to improve strains in order to address the financial problems.
Many genes involved in the Kennedy (TAG) pathway and fatty acid synthesis were overexpressed, as were the pathways involved in the carbohydrate synthesis, to boost the production of lipids [34]. A large number of efforts have been made in recent decades to increase the use of cyanobacteria and microalgae by metabolic, synthetic, and genetic engineering. Due to a lack of resources, bioinformatics, and multi-omics databases, photoautotrophic cell factories have rarely developed to the same level as their heterotrophic counterparts [36].
The genomes of a few microalgae, including C. reinhardtii CC503, Chlorella variabilis NC64, and Chlorella sorokiniana UTEX1602 [37], have been fully sequenced. This has established the basis for the genetic alteration of microalgae. Over the past ten years, short palindromic repeats that are clustered regularly interspaced and have an associated protein 9 (CRISPR/Cas9) have proven the most inventive and successful method [38]. CRISPR technology has been used in a variety of ways to improve cellular metabolism, regulate biosynthetic pathways, and increase the rate of production of metabolites. Using model microalgae C. reinhardtii, the first report of CRISPR-Cas9 gene editing was published in 2014 [37]. Prokaryotic cyanobacteria, such as Nostoc sp. PCC 7120, Synechococcus strains UTEX 2973 PCC 6301, PCC 7002, Synechocystis strains PCC 6803, have a relatively small genome and have been fully sequenced. They also have a higher growth rate and significantly higher lipid content. Therefore, compared to eukaryotic microalgae, they offer a far simpler operation system for genetic engineering, and numerous attempts have been made to boost the production of fatty acids and other green compounds [39]. Over the past few decades, researchers have devised a number of methods to increase the rate of biochemical production, either by genetically altering the genome of microbes or by overexpressing proteins.
Conclusion
Numerous problems arise from the large amounts of wastewater generated as a result of widespread water use for industrial, agricultural, and urban uses. The removal of contaminants via biological treatment frequently involves the use of bacteria, fungi, and algae. The use of algae in wastewater
treatment is more beneficial since it is used in treating pollutants and producing biomass as well. The contaminants are the only source of food and
energy that the algae can use to develop and create biomass. The algal biomass could be utilized for animal feed, liquid fuel production, composting,
and methane production. The problem of the scaleup procedure still has to be resolved, even with the optimistic research that has been published for
algae growth in wastewater and simultaneous micronutrient extraction. Further studies should focus more on genetic engineering and bioinformatics-based advances.
References
1. Awewomom J, Dzeble F, Takyi Y D, Ashie W B, Ettey E N Y O, Afua P E, Sackey L N, Opoku F, Akoto O. Addressing global environmental pollution using environmental control techniques: a focus on environmental policy and preventive environmental management. Discover Environment. 2024, 2 (1), 8. DOI: http://doi.org/10.1007/s44274-024-00033-5.
2. Malith Premaratne G K S H N, Randeniya Arachchige Don Praveen Anthonio, Vinoj Chamilka Liyanaarachchi, Bavatharny Thevarajah, P.H.V. Nimarshana, Anushree Malik, Thilini U. Ariyadasa,. Resource recovery from waste streams for production of microalgae biomass: A sustainable approach towards high-value biorefineries,. Bioresource Technology Reports,. 2022, 18, DOI: http://doi.org/https://doi.org/10.1016/j.biteb.2022.101070.
3. Aparicio J D, Raimondo E E, Saez J M, CostaGutierrez S B, Alvarez A, Benimeli C S, Polti M A. The current approach to soil remediation: a review of physicochemical and biological technologies, and the potential of their strategic combination. Journal of Environmental Chemical Engineering. 2022, 10 (2), 107141. DOI: http://doi.org/10.1016/j.jece.2022.107141.
4. Cai T, Park S Y, Li Y. Nutrient recovery from wastewater streams by microalgae: status and prospects. Renewable and Sustainable Energy Reviews. 2013, 19, 360-369. DOI: http://doi.org/10.1016/j.rser.2012.11.030.
5. Goswami R K, Agrawal K, Verma P. Multifaceted role of microalgae for municipal wastewater treatment: a futuristic outlook toward wastewater management. CLEANSoil, Air, Water. 2023,51(3), DOI: http://doi.org/10.1002/clen.202100286.
6. Verasoundarapandian G, Lim Z S, Radziff S B M, Taufik S H, Puasa N A, Shaharuddin N A, Merican F, Wong C Y, Lalung J, Ahmad S A. Remediation of pesticides by microalgae as feasible approach in agriculture: bibliometric strategies. Agronomy. 2022, 12 (1), 117. DOI: http://doi.org/10.3390/agronomy12010117.
7. Bhatt P, Bhandari G, Bhatt K, Simsek H. Microalgae-based removal of pollutants from wastewaters: Occurrence, toxicity and circular economy. Chemosphere. 2022, 306, 135576. DOI: http://doi.org/10.1016/j.chemosphere.2022.135576.
8. Di Lena G, Casini I, Lucarini M, Lombardi-Boccia G. Carotenoid profiling of five microalgae species from large-scale production. Food research
international. 2019, 120, 810-818. DOI: http://doi.org/10.1016/j.foodres.2018.11.043.
9. Al-Jabri H, Das P, Khan S, Thaher M, AbdulQuadir M. Treatment of wastewaters by microalgae and the potential applications of the produced biomass a review. Water. 2020, 13 (1), 27. DOI: http://doi.org/10.3390/w13010027.
10. Hachicha R, Elleuch J, Dubessay P, Hachicha R, Abdelkafi S, Michaud P, Fendri I. Integrated processes for olive mill wastewater treatment and its revalorization for microalgae culture. International microbiology : the official journal of the Spanish Society for Microbiology. 2025, 28 (5), 1071-1085. DOI: http://doi.org/10.1007/s10123-024-00600-z.
11. Monteiro dos Santos L, Barbosa da Silva J C, de Farias Silva C E, Villar da Gama B M, Almeida Medeiros J, Markou G, Rosas Garcia Almeida R M, Abud A K D S. Co-Cultivation between the Microalga Tetradesmus obliquus and Filamentous Fungus Cunninghamella echinulata Improves
Tertiary Treatment of Cheese Whey Effluent in Semicontinuous Mode. Processes. 2024, 12 (8), DOI: http://doi.org/10.3390/pr12081573.
12. Wang J, Tian Q, Zeng W, Qiu G, Shen L. Insights about fungus-microalgae symbiotic system in microalgae harvesting and wastewater treatment: A
review. Renewable and Sustainable Energy Reviews. 2023, 182, DOI: http://doi.org/10.1016/j.rser.2023.113408.
13. Ferreira A, Reis A, Vidovic S, Vladic J, Gkelis S, Melkonyan L, Avetisova G, Congestri R, Acién G, Muñoz R, Collet P, Combining microalgae based wastewater treatment with biofuel and biobased production in the frame of a biorefinery. In Grand Challenges in Algae Biotechnology, 2019; pp
319-369.
14. Chen L, Zhang X, Zhang M, Zhu Y, Zhuo R. Removal of heavy-metal pollutants by white rot fungi: Mechanisms, achievements, and perspectives. Journal of Cleaner Production. 2022, 354, 131681. DOI: http://doi.org/10.1080/21501203.2024.2389290.
15. Arakawa S, Kanaseki T, Wagner R, Goodenough U. Ultrastructure of the foliose lichen Myelochroa leucotyliza and its solo fungal and algal (Trebouxia
sp.) partners. Algal Research. 2022, 62, 102571. DOI: http://doi.org/10.1016/j.algal.2021.102571.
16. Qixin L, Xuan F, Zhiya S, Wenxin S, Shuo W, Ji L. Enhanced wastewater treatment performance by understanding the interaction between algae and bacteria based on quorum sensing. Bioresource technology. 2022, 354, 127161. DOI: http://doi.org/10.1016/j.biortech.2022.127161.
17. Chojnacka K, Moustakas K, Witek-Krowiak A. Bio-based fertilizers: A practical approach towards circular economy. Bioresource technology. 2020,295, 122223. DOI: http://doi.org/10.1016/j.biortech.2019.122223.
18. Carrillo V, Fuentes B, Gómez G, Vidal G. Characterization and recovery of phosphorus from wastewater by combined technologies. Reviews in Environmental Science and Bio/Technology. 2020,19, 389-418. DOI: http://doi.org/10.1007/s11157-020-09533-1.
19. Lam T P, Lee T M, Chen C Y, Chang J S. Strategies to control biological contaminants during microalgal cultivation in open ponds. Bioresource technology. 2018, 252, 180-187. DOI: http://doi.org/10.1016/j.biortech.2017.12.088.
20. Zhou W, Chen P, Min M, Ma X, Wang J, Griffith R, Hussain F, Peng P, Xie Q, Li Y, Shi J. Environment-enhancing algal biofuel production using wastewaters. Renewable and Sustainable Energy Reviews. 2014, 36, 256-269. DOI: http://doi.org/10.1016/j.rser.2014.04.073.
21. Arora K, Kaur P, Kumar P, Singh A, Patel S K S, Li X, Yang Y H, Bhatia S K, Kulshrestha S. Valorization of wastewater resources into biofuel and value-added products using microalgal system. Frontiers in Energy Research. 2021, 9, 646571. DOI: http://doi.org/10.3389/fenrg.2021.646571.
22. Al-Juboori R A, Al-Shaeli M, Aani S A, Johnson D, Hilal N. Membrane Technologies for Nitrogen Recovery from Waste Streams: Scientometrics and Technical Analysis. Membranes. 2022, 13 (1), DOI: http://doi.org/10.3390/membranes13010015.
23. Pang Y, Wang J. Various electron donors for biological nitrate removal: A review. Science of the Total Environment. 2021, 794, DOI:
http://doi.org/10.1016/j.scitotenv.2021.148699.
24. Mishra B K, Rastogi A, Shukla S, Regulatory role of mineral elements in the metabolism of medicinal plants. In Mineral Nutrition of Medicinal and
Aromatic Plants, Medicinal and Aromatic Plant Science and Biotechnology, 2012; Vol. 6, pp 1-23.
25. Nguyen L N, Aditya L, Vu H P, Johir A H, Bennar L, Ralph P, Hoang N B, Zdarta J, Nghiem L D. Nutrient removal by algae-based wastewater treatment. Current Pollution Reports. 2022, 8 (4), 369-383. DOI: http://doi.org/10.1007/s40726-022-00232-6.
26. Badar S N, Mohammad M, Emdadi Z, Yaakob Z. Algae and their growth requirements for bioenergy: A review. Biofuels. 2021, DOI: http://doi.org/10.1080/17597269.2018.1472978.
27. Song H, Li J, Su Q, Li H, Guo X, Shao S, Fan L, Xu P, Zhou W, Qian J. Insight into the mechanism of nitrogen sufficiency conversion strategy for microalgae-based ammonium-rich wastewater treatment. Chemosphere. 2024, 349, 140904. DOI: http://doi.org/10.1016/j.chemosphere.2023.140904.
28. Wehrs M, Tanjore D, Eng T, Lievense J, Pray T R, Mukhopadhyay A. Engineering Robust Production Microbes for Large-Scale Cultivation. Trends in microbiology. 2019, 27 (6), 524-537. DOI: http://doi.org/10.1016/j.tim.2019.01.006.
29. Sproles A E, Fields F J, Smalley T N, Le C H, Badary A, Mayfield S P. Recent advancements in the genetic engineering of microalgae. Algal Research. 2021, 53, DOI: http://doi.org/10.1016/j.algal.2020.102158.
30. Tran N T, Kaldenhoff R. Achievements and challenges of genetic engineering of the model green alga Chlamydomonas reinhardtii. Algal Research. 2020, 50, DOI: http://doi.org/10.1016/j.algal.2020.101986.
31. Daneshvar E, Wicker R J, Show P L, Bhatnagar A. Biologically-mediated carbon capture and utilization by microalgae towards sustainable CO2 biofixation and biomass valorization A review. Chemical Engineering Journal. 2022, 427, 130884. DOI: http://doi.org/10.1016/j.cej.
32. Siddiki S Y A, Mofijur M, Kumar P S, Ahmed S F, Inayat A, Kusumo F, Badruddin I A, Khan T Y, Nghiem L D, Ong H C, Mahlia T M I. Microalgae biomass as a sustainable source for biofuel, biochemical and biobased value-added products: An integrated biorefinery concept. Fuel. 2022, 307, DOI: http://doi.org/10.1016/j.fuel.2021.121782.
33. Kuo C M, Sun Y L, Lin C H, Lin C H, Wu H T, Lin C S. Cultivation and biorefinery of microalgae (Chlorella sp.) for producing biofuels and other byproducts: A review. Sustainability. 2021, 13 (23), DOI: http://doi.org/10.3390/su132313480.
34. Shahid A, Malik S, Zhu H, Xu J, Nawaz M Z, Nawaz S, Asraful Alam M, Mehmood M A. Cultivating microalgae in wastewater for biomass production, pollutant removal, and atmospheric carbon mitigation; a review. The Science of the total environment. 2020, 704, 135303. DOI: http://doi.org/10.1016/j.scitotenv.2019.135303.
35. Rahman K M, Food and high value products from microalgae: market opportunities and challenges. In Microalgae Biotechnology for Food, Health and High Value Products, 2020; pp 3-27.
36. Kumari R, Patel V K, Kumari P, Kajla S, Yield Enhancement in Algal Production and Genetic Engineering Tools for Microalgal Biorefinery. In Value Added Products From Bioalgae Based Biorefineries: Opportunities and Challenges, Springer Nature Singapore: 2024; pp 489-522.
37. Shin S E, Koh H G, Kang N K, Suh W I, Jeong B R, Lee B, Chang Y K. Isolation, phenotypic characterization and genome wide analysis of a Chlamydomonas reinhardtii strain naturally modified under laboratory conditions: towards enhanced microalgal biomass and lipid production for biofuels. Biotechnology for biofuels. 2017,10, 308. DOI: http://doi.org/10.1186/s13068-017-1000-0.
38. Rao Y, Yang X, Pan C, Wang C, Wang K. Advance of Clustered Regularly Interspaced Short Palindromic Repeats-Cas9 System and Its Application in Crop Improvement. Frontiers in plant science. 2022, 13, 839001. DOI: http://doi.org/10.3389/fpls.2022.839001.
39. Santos-Merino M, Singh A K, Ducat D C. New Applications of Synthetic Biology Tools for Cyanobacterial Metabolic Engineering. Frontiers in bioengineering and biotechnology. 2019, 7, 33. DOI: http://doi.org/10.3389/fbioe.2019.00033.
Open Access
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third-party material are included in the article’s Creative Common license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Common’s license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
Creative Commons
This is an open access article distributed under the terms of the Creative Commons CC BY license, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. You are not required to obtain permission to reuse this article. To request permission for a type of use not listed, please contact Biotechnology Kiosk.